Home / Education Hub
Geographic atrophy
education hub
Get helpful resources for your practice designed to
help you make a difference
Watch and Discover
See how GA progresses over time and how it may affect a patient’s vision, daily activities, and independence.
GA by the Numbers
GA Is an Irreversible
Cause of Vision Loss1,2

Drusen Are a Hallmark of Early and Intermediate Disease
Drusen are a hallmark of early and intermediate AMD, which can be observed by direct examination on color fundus photography (CFP) or on optical coherence tomography (OCT).1,2,5,6 Drusen come in various sizes. The larger the drusen, the greater the chance of progression to an advanced form of AMD such as GA.1, 2
Early AMD with drusen (white arrows). Pigmentary abnormalities are absent. Drusen are found between the retinal pigment epithelium basement membrane and Bruch’s membrane.3
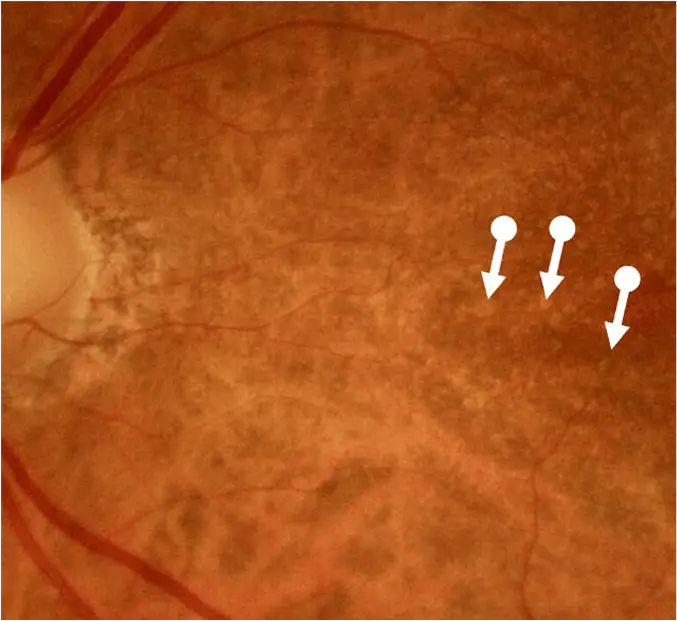
Image courtesy of Dr. Mohammad Rafieetary
Degenerative Changes Occur in
Intermediate Disease
According to the Beckman Classification, early AMD is characterized by a few, small drusen measuring >63 and ≤ 124 µm without pigmentary changes. Intermediate AMD is defined by the presence of at least one large drusen >125 µm, with or without pigmentary changes. Advanced AMD includes the development of either geographic atrophy (GA) or choroidal neovascularization (CNV).3,4
Importantly, changes in visual function can occur before measurable declines in visual acuity, and patients should be advised to promptly report any sudden or persistent changes in vision, such as blurriness or distortion.7,8
Lesion Areas Enlarge in GA
Dry AMD progression to GA is characterized by the development of new atrophic lesions, growth of individual areas, foveal encroachment, or coalescence of multiple lesions. GA can be detected using various imaging modalities, such as OCT and fundus autofluorescence (FAF), which are commonly available in most clinics.1-3,9
Although less sensitive for detecting early GA, color fundus photography can reveal GA lesions as hypopigmented areas with sharply demarcated borders. Fundus autofluorescence (FAF) is one of the primary modalities for detecting and monitoring GA. On FAF imaging, GA lesions appear as well-defined hypofluorescent (dark) areas due to loss of the retinal pigment epithelium (RPE). On OCT, characteristic features of GA include loss of the photoreceptors, loss of the RPE, and areas of choroidal hypertransmission, which appear as bright vertical columns with increased visualization of the underlying choroidal structures.1,3,4
Monitoring for Progression Is Critical
Lesion patterns can be predictive of slower or faster progressing disease and provide key data to inform management strategies.2,3 Patients can present with a wide range of visual symptoms; therefore it is critical to monitor patients for disease progression.4,7
Lesion Size
Slower
Progression

Small
Baseline Lesions
Faster
Progression

Large
Baseline Lesions
Location
Slower
Progression

Foveal
Faster
Progression

Non-Foveal
Focality
Slower
Progression

Unifocal
Faster
Progression

Multifocal
More Than You Realize
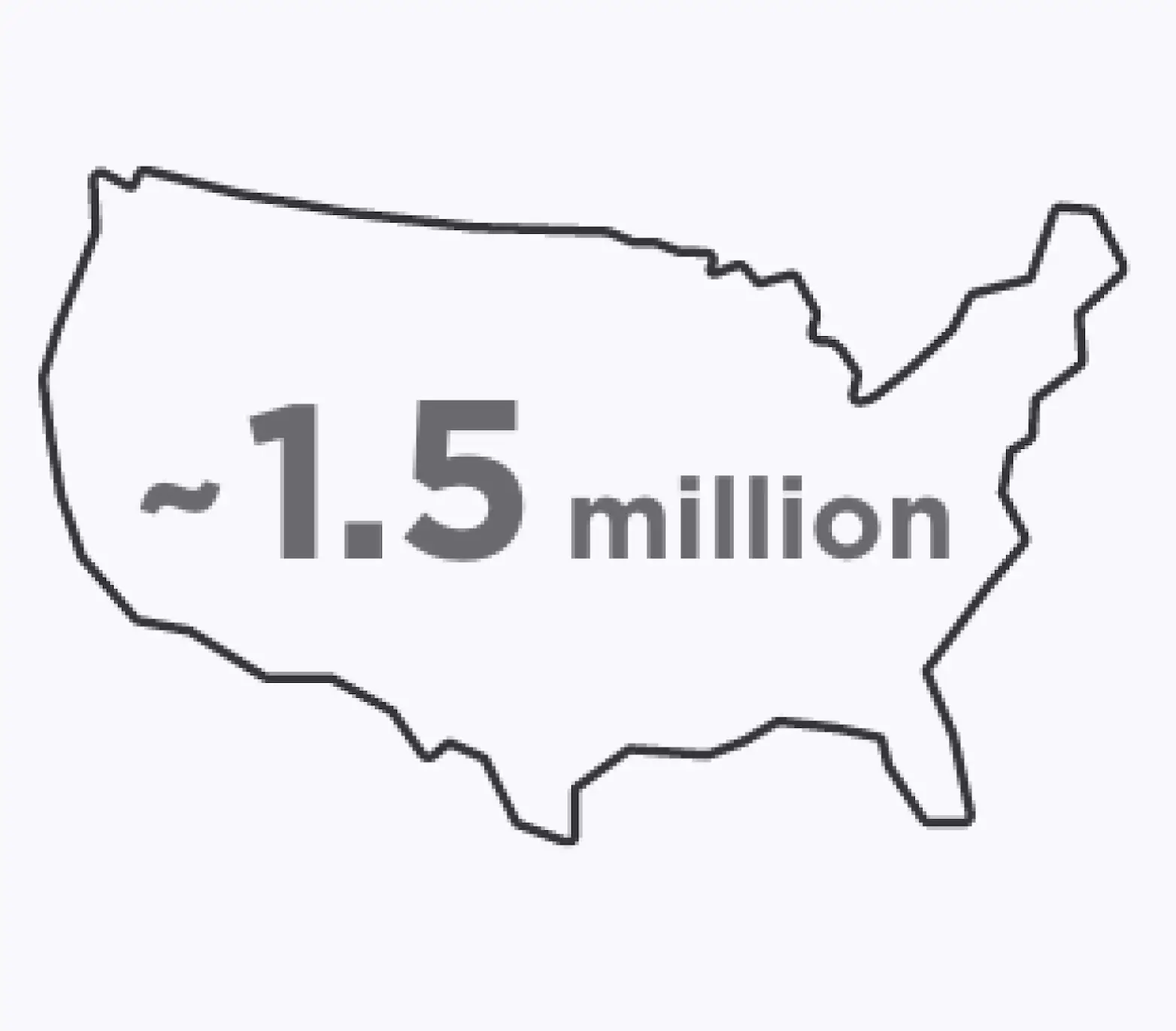
people in the United
States are known to have GA11


of cases of legal
blindness are attributed to GA in North America10
*GA progression is different for everyone. Some people may experience slower or faster GA progression and changes in vision.
Fear of Vision Loss
Patients Suffer From


Central field defect14
Vision Loss7
Leads To
What Patients Worry About
Missing meaningful life
events13, 15-17

Difficulties with activities
of daily life17, 18
Loss of independence
(e.g. difficulty with
driving)13, 16-19
An Expanding Population
Advanced Age is Not the Only Risk Factor 7

Caucasian descent7




Visual Acuity ≠ Visual Function
Monitoring for progression can be difficult since disease progression is NOT always correlated with a decline in visual acuity, particularly with extrafoveal lesions.4
To get a complete look at how geographic atrophy (GA) is impacting your patients, you can go beyond best corrected visual acuity (BCVA) by evaluating functional assessments.4
Functional Tests to Assess GA
Contrast Sensitivity
Low Light Visual Acuity (LLVA)
Dark Adaptation
Delayed dark adaptation may provide evidence of AMD before the appearance of clinical features such as drusen and focal pigmentary changes10,20
Microperimetry
GA Changes Lives
Patients Lose More Than Just Vision

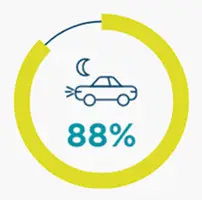
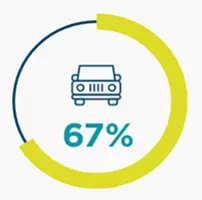
*67% became ineligible to drive with a median time to progression of 1.6 years (IQR, 0.7-2.7) from the index date.
Watch and Discover
Learn how the complement system — including both C3 and C5 — plays a role in the progression of Geographic Atrophy (GA).
Understanding the Complement Pathway
The complement system acts as the first line of defense against infection and plays a central role in the immune response. Dysregulation of this pathway — particularly at the C3 and C5 levels — has been associated with GA progression.2,22
Targeting C3 or C5 within the complement cascade may offer different therapeutic approaches, each with potential benefits and limitations.22
The chart below illustrates key components of the complement system and how they relate to Geographic Atrophy.
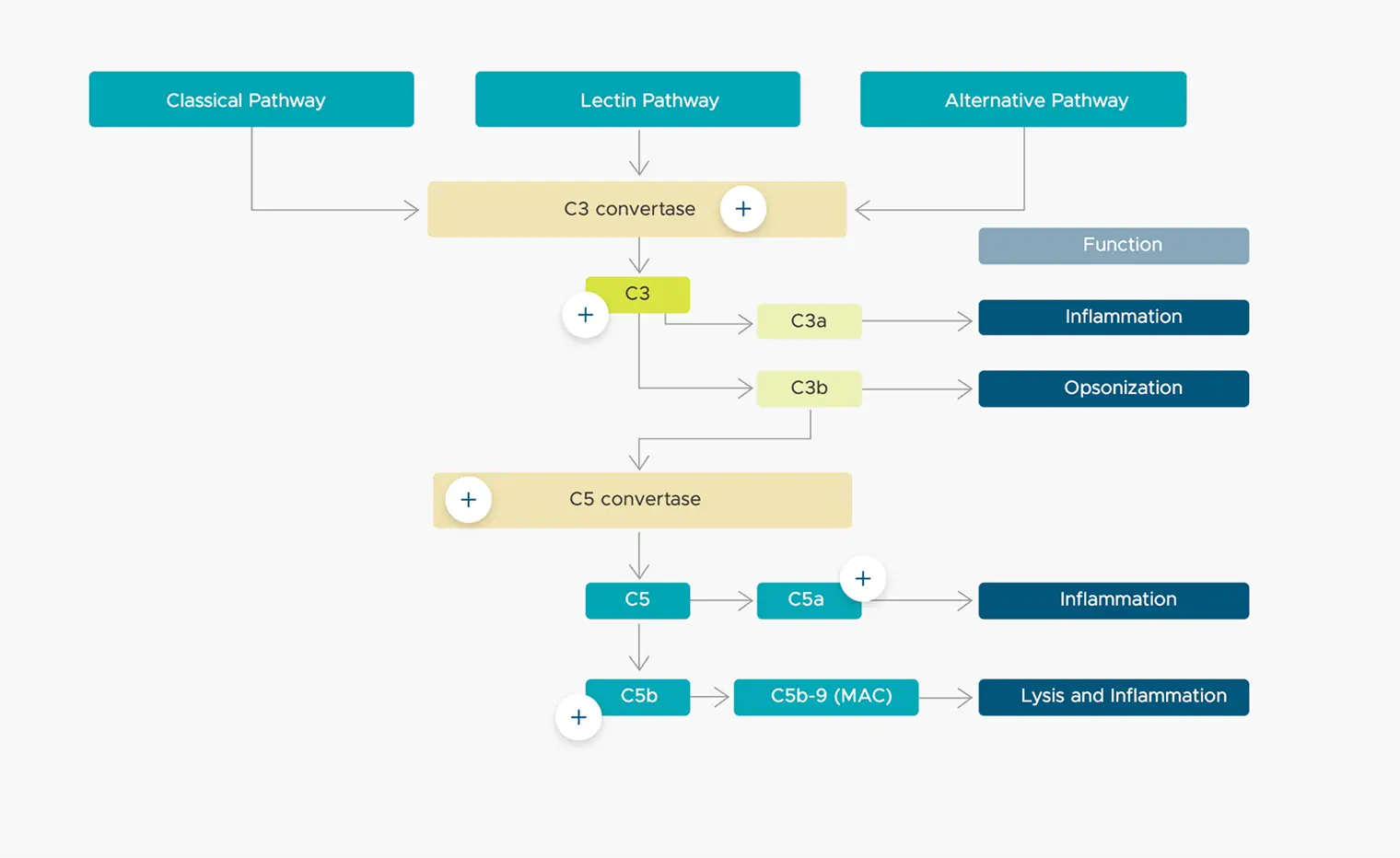
All 3 complement pathways converge
at the C3 convertase enzyme2
C3 convertase cleaves C3 to form
C3a, which has proinflammatory
effects through inflammasome activation,
and C3b, which plays a role in pathogen
opsonization and can be beneficial for
anti-inflammatory activities2,22,23
As C5 convertase cleaves C5 into C5a
and C5b, it initiates the terminal cascade
of the compliment system2
C5a
Can lead to inflammation
causing death of retinal pigment
epithelial (RPE) cells2,24,25
Identifying GA
Watch and Discover
See how GA progresses over time and how it may affect a patient’s vision, daily activities, and independence.
GA by the Numbers
GA Is an Irreversible
Cause of Vision Loss1,2

Drusen Are a Hallmark of Early and Intermediate Disease
Drusen are a hallmark of early and intermediate AMD, which can be observed by direct examination on color fundus photography (CFP) or on optical coherence tomography (OCT).1,2,5,6 Drusen come in various sizes. The larger the drusen, the greater the chance of progression to an advanced form of AMD such as GA.1, 2

Image courtesy of Dr. Mohammad Rafieetary
Early AMD with drusen (white arrows). Pigmentary abnormalities are absent. Drusen are found between the retinal pigment epithelium basement membrane and Bruch’s membrane.3
Degenerative Changes Occur in
Intermediate Disease
According to the Beckman Classification, early AMD is characterized by a few, small drusen measuring >63 and ≤ 124 µm without pigmentary changes. Intermediate AMD is defined by the presence of at least one large drusen >125 µm, with or without pigmentary changes. Advanced AMD includes the development of either geographic atrophy (GA) or choroidal neovascularization (CNV).3,4
Importantly, changes in visual function can occur before measurable declines in visual acuity, and patients should be advised to promptly report any sudden or persistent changes in vision, such as blurriness or distortion.7,8
Lesion Areas Enlarge in GA
Dry AMD progression to GA is characterized by the development of new atrophic lesions, growth of individual areas, foveal encroachment, or coalescence of multiple lesions. GA can be detected using various imaging modalities, such as OCT and fundus autofluorescence (FAF), which are commonly available in most clinics.1-3,9
Although less sensitive for detecting early GA, color fundus photography can reveal GA lesions as hypopigmented areas with sharply demarcated borders. Fundus autofluorescence (FAF) is one of the primary modalities for detecting and monitoring GA. On FAF imaging, GA lesions appear as well-defined hypofluorescent (dark) areas due to loss of the retinal pigment epithelium (RPE). On OCT, characteristic features of GA include loss of the photoreceptors, loss of the RPE, and areas of choroidal hypertransmission, which appear as bright vertical columns with increased visualization of the underlying choroidal structures.1,3,4
Monitoring for Progression Is Critical
Lesion patterns can be predictive of slower or faster progressing disease and provide key data to inform management strategies.2,3 Patients can present with a wide range of visual symptoms; therefore it is critical to monitor patients for disease progression.4,7
Lesion Size
Slower
Progression

Small
Baseline Lesions
Faster
Progression

Large
Baseline Lesions
Location
Slower
Progression

Foveal
Faster
Progression

Non-Foveal
Focality
Slower
Progression

Unifocal
Faster
Progression

Multifocal
The Current Challenge
More Than You Realize
Age-related macular degeneration (AMD) can advance to geographic atrophy (GA), which can cause irreversible vision loss.2,10

people in the United
States are known to have GA11


of cases of legal
blindness are attributed to GA in North America10
*GA progression is different for everyone. Some people may experience slower or faster GA progression and changes in vision.
Fear of Vision Loss
Patients Suffer From


Central field defect14

Vision Loss7
Which
Leads To
What Patients Worry About

Missing meaningful life
events13, 15-17

Difficulties with activities
of daily life17, 18

Loss of independence
(e.g. difficulty with
driving)13, 16-19
An Expanding Population
People who are 55 years of age and older are at a greater risk of developing GA. Along with baby boomers, the aging Generation X will start to become susceptible to GA.7
Advanced Age is Not the Only Risk Factor 7

Caucasian descent7

Hypertension7 / cardiovascular
disease7


Low levels of systemic antioxidants7

Impact on Patients
Visual Acuity ≠ Visual Function
Monitoring for progression can be difficult since disease progression is NOT always correlated with a decline in visual acuity, particularly with extrafoveal lesions.4
To get a complete look at how geographic atrophy (GA) is impacting your patients, you can go beyond best corrected visual acuity (BCVA) by evaluating functional assessments.4
Functional Tests to Assess GA
Contrast Sensitivity
Low Light Visual Acuity (LLVA)
Dark Adaptation
Delayed dark adaptation may provide evidence of AMD before the appearance of clinical features such as drusen and focal pigmentary changes10,20
Microperimetry
GA Changes Lives
Geographic atrophy (GA) can have an impact on patients’ emotional well-being, leading to feelings of anger, frustration, isolation, fear, and depression.13,18,19
Patients Lose More Than Just Vision



*67% became ineligible to drive with a median time to progression of 1.6 years (IQR, 0.7-2.7) from the index date.
The Complement Pathway
Watch and Discover
Learn how the complement system — including both C3 and C5 — plays a role in the progression of Geographic Atrophy (GA).
Understanding the Complement Pathway
The complement system acts as the first line of defense against infection and plays a central role in the immune response. Dysregulation of this pathway — particularly at the C3 and C5 levels — has been associated with GA progression.2,22
Targeting C3 or C5 within the complement cascade may offer different therapeutic approaches, each with potential benefits and limitations.22
The chart below illustrates key components of the complement system and how they relate to Geographic Atrophy.
Targeting complement by inhibiting C3 or C5 within the pathway may interrupt processes driving GA.2,22
C3 Convertase
All 3 complement pathways converge at the C3 convertase enzyme2
C3
C3 convertase cleaves C3 to form C3a, which has proinflammatory effects through inflammasome activation, and C3b, which plays a role in pathogen opsonization and can be beneficial for anti-inflammatory activities2,22,23
C5 Convertase
As C5 convertase cleaves C5 into C5a and C5b, it initiates the terminal cascade of the compliment system2
C5a
Can lead to inflammation causing death of retinal pigment epithelial (RPE) cells2,24,25
C5b
References
- Holz FG, Schmitz-Valckenberg S, Fleckenstein M. Recent developments in the treatment of age-related macular degeneration. J Clin Invest. 2014;124(4):1430-1438.
- Boyer DS, Schmidt-Erfurth U, van Lookeren Campagne M, Henry EC, Brittain C. The pathophysiology of geographic atrophy secondary to age-related macular degeneration and the complement pathway as a therapeutic target. Retina. 2017;37(5):819-835.
- Fleckenstein M, Mitchell P, Freund KB, et al. The progression of geographic atrophy secondary to age-related macular degeneration. Ophthalmology. 2018;125(3):369-390.
- Sadda SR, Chakravarthy U, Birch DG, et al. Clinical endpoints for the study of geographic atrophy secondary to age-related macular degeneration. Retina. 2016;36(10):1806-1822.
- Monés J, Garcia M, Biarnés M, Lakkaraju A, Ferraro L. Drusen ooze: a novel hypothesis in geographic atrophy. Ophthalmol Retina. 2017;1(6):461-473.
View More
- Ambati J, Ambati BK, Yoo SH, Ianchulev S, Adamis AP. Age-related macular degeneration: etiology, pathogenesis, and therapeutic strategies. Surv Ophthalmol. 2003;48(3):257-293.
- Flaxel CJ, Adelman RA, Bailey ST, et al. Age-related macular degeneration preferred practice pattern(R). Ophthalmology. 2020;127(1):P1-P65.
- Sunness JS, Rubin GS, Applegate CA, et al. Visual function abnormalities and prognosis in eyes with age-related geographic atrophy of the macula and good visual acuity. Ophthalmology. 1997;104(10):1677-1691.
- Lindblad AS, Lloyd, PC, Clemons TE, et al. Change in area of geographic atrophy in the Age-Related Eye Disease Study: AREDS report number 26. Arch Ophthalmol. 2009;127(9):1168-1174.
- Holz FG, Strauss EC, Schmitz-Valckenberg S, van Lookeren Campagne M. Geographic atrophy: clinical features and potential therapeutic approaches. Ophthalmology. 2014;121(5):1079-1091.
- Desai D, Dugel PU. Complement cascade inhibition in geographic atrophy: a review. Eye (Lond). 2022;36(2):294-302.
- Bakri SJ, Bektas M, Sharp D, Luo R, Sarda SP, Khan S. Geographic atrophy: mechanism of disease, pathophysiology, and role of the complement system. J Manag Care Spec Pharm. 2023;29(5-a Suppl):S2-S11.
- Carlton J, Barnes S, Haywood A. Patient perspectives in geographic atrophy (GA): exploratory qualitative research to understand the impact of GA for patients and their families. Br Ir Orthopt J. 2019;15(1):133-141.
- Stahl A. The diagnosis and treatment of age-related macular degeneration. Dtsch Arztebl Int. 2020;117(29-30):513-520.
- Sayegh RG, Sacu S, Dunavölgyi R, et al. Geographic atrophy and foveal-sparing changes related to visual acuity in patients with dry age-related macular degeneration over time. Am J Ophthalmol. 2017;179:118-128.
- Chakravarthy U, Bailey CC, Johnston RL, et al. Characterizing disease burden and progression of geographic atrophy secondary to age-related macular degeneration. Ophthalmology. 2018;125(6):842-849.
- Patel PJ, Ziemssen F, Ng E, et al. Burden of illness in geographic atrophy: a study of vision-related quality of life and health care resource use. Clin Ophthalmol. 2020;14:15-28.
- Singh RP, Patel SS, Nielsen JS, Schmier JK, Rajput Y. Patient-, caregiver-, and eye care professional-reported burden of geographic atrophy secondary to age-related macular degeneration. Am J Ophthalmic Clin Trials. 2019;2(1):1-6.
- Sivaprasad S, Tschosik EA, Guymer RH, et al. Living with geographic atrophy: an ethnographic study. Ophthalmol Ther. 2019;8(1):115-124.
- Higgins BE, Taylor DJ, Binns AM, Crabb DP. Are current methods of measuring dark adaptation effective in detecting the onset and progression of age-related macular degeneration? A systematic literature review. Ophthalmol Ther. 2021;10(1):21-38.
- Meleth AD, Mettu P, Agron E, et al. Changes in retinal sensitivity in geographic atrophy progression as measured by microperimetry. Invest Ophthalmol Vis Sci. 2011;52(2):1119-1126.
- Xu H, Chen M. Targeting the complement system for the management of retinal inflammatory and degenerative diseases. Eur J Pharmacol. 2016;787:94-104.
- Coulthard LG, Woodruff TM. Is the complement activation product C3a a proinflammatory molecule? Re-evaluating the evidence and the myth. J Immunol. 2015;194(8):3542-3548.
- Xie CB, Jane-Wit D, Pober JS. Complement membrane attack complex: new roles, mechanisms of action, and therapeutic targets. Am J Pathol. 2020;190(6):1138-1150.
- Brandstetter C, Holz FG, Krohne TU. Complement component C5a primes retinal pigment epithelial cells for inflammasome activation by lipofuscin-mediated photooxidative damage. J Biol Chem. 2015;290(52):31189-31198.
- Kumar-Singh R. The role of complement membrane attack complex in dry and wet AMD – from hypothesis to clinical trials. Exp Eye Res. 2019;184:266-277.





